
Loading...

Loading...
Atoms were believed by early Indian and Greek philosophers to be indivisible, elementary components of all substances. In ancient Babylonia (between the 18th and 16th centuries BCE) five classical elements were described: sea, earth, fire, sky, and wind. The classical Greek elements (earth, water, air, fire, and sometimes the aether) date from before 400 BCE and persisted throughout the Middle Ages and into the Renaissance, deeply influencing European thought and culture. According to Aristotle in his On Generation and Corruption:
By the 17th and 18th centuries CE, chemists showed that this model was totally incorrect, but that certain substances could not be further broken down by chemical methods; these constitute our current understanding of what an atom is. During the late 19th and early 20th centuries, physicists discovered subatomic components and structure inside the atom, thereby demonstrating that the atom is divisible. The principles of quantum mechanics were used to successfully model the atom (see my description in explaining the Big Bang).
Chemists were, by the 19th century, discovering and classifying dozens of atoms, and they even used solar spectrometry to discover helium on the Sun before it had been identified on Earth. They also realized that the best way of explaining atoms was to imagine them as tiny ‘solar systems’ with charged small particles orbiting a heavier nucleus.

The atom is a basic unit of matter that consists of a dense central nucleus surrounded by a cloud of negatively charged electrons. The nucleus contains positively charged protons and electrically neutral neutrons (except in the case of hydrogen-1, which is the only stable atomic nucleus with no neutrons). The electrons of an atom are bound to the nucleus by the electromagnetic force.
A group of atoms can remain bound to each other, forming a molecule. An atom containing an equal number of protons and electrons is electrically neutral, otherwise it has a positive or a negative charge depending on the difference in the number of electrons. A positively or negatively charged atom is known as an ion. An atom is classified according to the number of protons and neutrons in its nucleus: the number of protons determines the chemical element, and the number of neutrons determines the isotope of the element.
Over 99.94% of an atom’s mass is concentrated in the nucleus, with protons and neutrons having roughly equal mass.
The electrons determine the chemical properties of an element, and strongly influence an atom’s magnetic properties.
In 1913, Niels Bohr suggested that the electrons were confined in clearly defined orbits or ‘shells’, and could jump between these, but could not freely spiral inward or outward in intermediate states. An electron must absorb or emit specific amounts of energy (a quantum of energy) to transfer between these fixed orbits.
In 1926, Erwin Schrödinger developed a mathematical model of the atom that described the electrons as three-dimensional waveforms rather than point particles. A consequence of using waveforms to describe particles is that it is mathematically impossible to obtain precise values for both the position and momentum of a particle at the same time; this is the uncertainty principle, formulated by Werner Heisenberg in 1926. It implies that, for a given accuracy in measuring a position, one could only obtain a range of probable values for momentum, and vice versa. This model was able to explain observations of atomic behaviour that previous models could not, such as certain structural and spectral patterns of atoms larger than hydrogen. Thus, the ‘planetary’ model of the atom was discarded in favour of one that described atomic orbital zones around the nucleus where a given electron is most likely to be observed.
Though the word atom originally denoted a particle that cannot be cut into smaller particles, in modern scientific usage the atom is composed of various subatomic particles. The principal constituent particles of an atom are the electron, the proton and the neutron.
The electron is by far the least massive of these particles at 9.11 × 10−31 kg, with a negative electrical charge and a size that is too small to be measured using available techniques.
Protons have a positive charge and a mass 1,836 times that of the electron, at 1.6726 × 10−27 kg.
Neutrons have no electrical charge and have a free mass of 1,839 times the mass of electrons. Neutrons and protons have comparable dimensions, of the order of 2.5 × 10−15 m, although the ‘surface’ of these particles is not sharply defined.




Protons, neutrons and electrons are not the end of the story; they consist of more primitive components, each of which has four quantum numbers; these numbers are labelled:
A fermion (named after Enrico Fermi) is any particle with half-odd-integer spin; no two fermions in the same atom may have the same four quantum numbers (this is the ‘Pauli exclusion principle’ [see below], named after Wolfgang Pauli).
For example, no two electrons in a single atom can have the same four quantum numbers; if n, ℓ, and mℓ are the same, ms must be different so that the electrons have opposite spins, and so on.
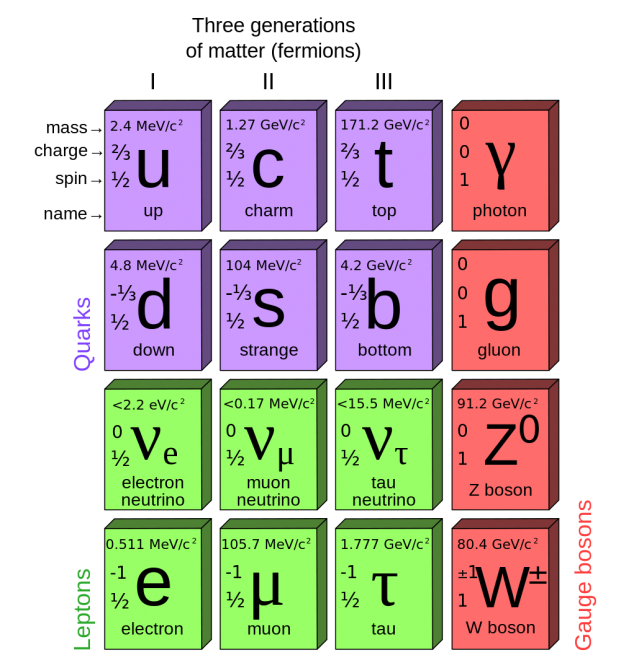
The Standard Model (the theoretical framework describing all the currently known elementary particles, and some unobserved ones) recognizes two types of elementary fermions: quarks and leptons. In all, the model distinguishes 24 different fermions: 6 quarks and 6 leptons, each with a corresponding anti-particle.
Fermions are contrasted with bosons. A fermion can be an elementary particle, such as the electron; or it can be a composite particle, such as the proton. Particles with integer (whole number) spin are bosons, while particles with half-integer spin are fermions.
Bosons, unlike fermions, are not subject to the Pauli exclusion principle: an unlimited number of particles may occupy the same state at the same time. This explains why, at low temperatures, bosons can behave very differently from fermions; all the particles will tend to congregate at the same lowest-energy state, forming what is known as a condensate. Composite fermions, such as protons and neutrons, are key building blocks of matter, whereas bosons are generally force carrier particles. As two fermions cannot occupy the same quantum state, matter has a “rigidity” or “stiffness”.
Bosons may be either elementary, like photons, or composite, like mesons. While most bosons are composite particles, there are six bosons which are elementary:
Unlike the gauge bosons and the Higgs boson, the graviton has not yet been observed experimentally.
Gauge bosons act as carriers of the fundamental forces of nature. More specifically, elementary particles exert forces on each other by the exchange of gauge bosons, usually as “virtual particles”. There are three kinds of gauge bosons:
The fourth fundamental interaction, gravity, may also be carried by a boson, called the graviton.
One expression of the Pauli Exclusion Principle is that “no two electrons in the same atom can be in the same quantum state.”. This means that no two electrons can have the same set of quantum states of energy, angular momentum magnitude, angular momentum orientation, and orientation of intrinsic spin.
Pauli’s Principle is based on the fact that any two given electrons are indistinguishable from one another and thus changing the designations between two or more electrons in different quantum states should have no observable effect. In describing the mathematical wave function of an atom — which is the product of the wave functions of the individual electrons — quantum physics mandates that the wave function itself (of the atom or electron) is also not observable. On the other hand, the squared magnitude of the atom’s wave function is observable, i.e. capable of being experimentally measured. Inasmuch as taking a square root of the squared magnitude of any given wave function will yield a plus (+) or minus (−) sign, the wave function of an atom or elementary particle can either be symmetric with respect to exchanging the designations of two of its constituent parts (the + sign), or anti-symmetric (the − sign).
The Pauli Exclusion Principle then specifies the wave functions of electrons, protons and other so-called spin-½ particles to be anti-symmetric. Thus when two electron designations are switched in the same atom or molecule, the total wave function of the atom or molecule changes sign.
This apparently esoteric principle is the very basis for the existence of the Periodic Table of the Elements. The Pauli Exclusion Principle allows for the distinction between different elements. Furthermore, the Principle also affects the way molecules form in the bonding of atoms, for the way molecules interact to form gases, liquids, or solids, and/or how the molecules aggregate themselves in living organisms. In effect, the Pauli Exclusion Principle “is the basis for the observable order of the universe, including the existence of life”. “Pauli’s Exclusion Principle accounts for atomic structure. Without this structure, every atom would collapse into a positive nucleus surrounded by an unorganized mass of negative charges.”
Perhaps, even more significantly, Pauli’s Exclusion Principle is not enforced by any physical force understood by mainstream science. “When an electron enters an ion, somehow it knows the quantum numbers of the electrons which are there, and somehow it knows which atomic orbitals it may enter, and which it may not”. This is nothing short of incredible! It implies consciousness or connectedness between any and all elementary particles, and by a method totally unknown to the mainstream purveyors of quantum physics.

In effect, an electron avoids occupied orbitals — in much the same way most motorists might avoid parking in occupied parking spaces — but this avoidance is not due to electrostatic repulsion or some mechanical property. The avoidance is due instead to the anti-symmetry requirement of the wave function of this spin-½ particle. This may be “another manifestation of the mind-like aspects of physical reality, similar to the power of information to affect observable physical states”.
When two atoms come within close proximity to one another, the concept of each being in a separate state loses its meaning. Thus two atoms with closed shells find they cannot form a chemical bond because the electrons in one atom find no available quantum states in the other in which to occupy. In effect, even though the atomic nuclei in molecules are surrounded by what is essentially empty space, the atoms cannot be forced together because the symmetry of the quantum wave functions forbid it. And the electrons know it! The electrons know what the other electrons are doing, and what quantum states they are in. And such knowing implies consciousness on the parts of elementary particles.
This potential for mindfulness becomes even more pronounced when we encounter the EPR Paradox (see below), where in David Bohm’s formulation two spin-½ particles form a ‘singlet state’, where the angular momenta of the two are counter-aligned in such a way as to cancel each other and yield a total angular momentum of zero.
Not only two atoms which have come in close proximity but any two quantum systems that have once interacted, as they are then separated over long distances they stay connected! Also, the knowingness involved implies an instantaneous means of communication. The knowledge contained in one particle being measured affects a second previously interacted particle at a distance. Locality is violated. Non-locality reigns and the speed of light is essentially ignored.
The Pauli Exclusion Principle is as fundamental a characteristic of Quantum Physics as there is. Albert Einstein and his colleagues Boris Poldolsky and Nathan Rosen attempted in their theoretical EPR Paradox to argue against the very viability of Quantum Physics. Quantum physics is fundamentally based on probabilities, whereas in Einstein’s view: “God does not play dice”. The EPR trio then attempted to show that either the Pauli Exclusion Principle was wrong (and thus Quantum Physics had failed in the most basic test of its validity), OR that the Special Relativity requirement that nothing can exceed the speed of light was in error. The latter ingredient of Relativity — known as locality — turned out to be the loser. The Pauli Exclusion Principle and Quantum Physics were shown experimentally to be the more valid concepts.
The Einstein-Poldolsky-Rosen Experiment (the EPR Experiment) has been described by several authors.
One describes the situation of two quantum particles which are once together flying apart and being measured at two distant locations. There exists a connection between the particles such that the fact of an observation of particle A is relayed to the distant particle B, in such a manner that the communication, “does not diminish with distance, cannot be shielded, and travels faster than light”. The fact of the two particles once being together is sufficient to mingle the particles’ phases (which the author refers to as quantum phase entanglement). This results in the effect being non-local (whereas all ordinary light-speed-limited forces are referred to as local). (There’s an interesting article on quantum entanglement in The Times of India.)
Another view of the EPR concept is to conceive of a pair of two particles travelling in opposite directions, and required by the Pauli Exclusion Principle to have opposite spins. (The Pauli Exclusion Principle may be considered to be absolutely essential to any viable theory of quantum mechanics, and in fact was the key link in which Einstein et al hoped to disprove the quantum theory. It just didn’t work out too well — the EPR Experiment has become a major supporting milestone for Quantum Physics.) Meanwhile when the spin of one particle is unilaterally changed, an astounding experimental result is that the second particle’s spin immediately flips of its own accord (and thus maintains the validity of the Pauli Exclusion Principle). And the means by which the information of the first spin flip is transferred to the second particle (so that it too can flip) is information which is required to travel faster than the speed of light. While the information transfer may not be simultaneous (limits on the experimental apparatus prohibits any proof of simultaneity), it nevertheless — within the time frame of the Planck constant or speeds in excess of the speed of light — must connect the two particles in some fundamental manner.

In 1964 Murray Gell-Mann and George Zweig suggested that hundreds of the subatomic particles known at the time could be explained as combinations of just three fundamental particles. Gell-Mann chose the name “quarks”, for these three particles, a nonsense word used by James Joyce in the novel Finnegan’s Wake: “Three quarks for Muster Mark!”. (After the fact, Gell-Mann was able to tack on the literary antecedent from Joyce, but the physicists’ quark was pronounced from the beginning to rhyme with cork.)
A quark is an elementary particle and a fundamental constituent of matter. Quarks combine to form composite particles called hadrons, the most stable of which are protons and neutrons, the components of atomic nuclei. Quarks are never directly observed or found in isolation; they can only be found within baryons or mesons. For this reason, much of what is known about quarks has been drawn from observations of the hadrons themselves.
The quark belongs to the fermion group of particles, which is one of the two basic constituents of matter (the other being the lepton, of which the electron is an example). There are, it turns out, six types of quarks, each having a fractional electric charge of either +⅔ or −⅓.
The six types of quarks (q), known as flavours: up (u), down (d), strange (s), charm (c), bottom (b), and top (t). (These names and letters have no inherent meaning; they were simply allocated as they were discovered, as ‘labels’ with which to refer to them.) Up and down quarks have the lowest masses of all quarks. The heavier quarks rapidly change into up and down quarks through a process of particle decay, the transformation from a higher mass state to a lower mass state. Because of this, up and down quarks are generally stable and the most common in the universe, whereas strange, charm, top, and bottom quarks can only be produced in high energy collisions (such as those involving cosmic rays and in particle accelerators).
Protons are composed of two up quarks and one down quark, while a neutron consists of one up quark and two down quarks. This distinction accounts for the difference in mass and charge between the two particles. The quarks are held together by the strong nuclear force, which is mediated by gluons. The gluon is a member of the family of gauge bosons, which are elementary particles that mediate (or govern) physical forces.
Quarks have various intrinsic properties, including electric charge, colour charge, mass, and spin. Quarks are the only elementary particles to experience all four fundamental forces (electromagnetism, gravitation, strong interaction, and weak interaction), as well as the only known particles whose electric charges are not integer multiples of the elementary charge. For every quark flavour there is a corresponding type of anti-particle, known as an antiquark, that has the same mass, mean lifetime, and spin as its respective quark, but the electric charge and other charges have the opposite sign. Antiquarks are denoted by a bar over the symbol for the corresponding quark, such as ū for an anti-up quark.
Unlike leptons, quarks possess colour charge, which causes them to engage in the strong interaction. The resulting attraction between different quarks causes the formation of composite particles known as hadrons.
Scientific American reported on 11th September 2015 that Two Accelerators Find Particles That May Break Known Laws of Physics, referring in detail to newly detected sub-atomic particles that appear to challenge the Standard Model of Elementary Particles – more Higgs Bosons perhaps?
The quarks which determine the quantum numbers of hadrons are called valence quarks; apart from these, any hadron may contain an indefinite number of virtual (or sea) quarks, antiquarks, and gluons which do not influence its quantum numbers. There are two families of hadrons: baryons, with three valence quarks, and mesons, with a valence quark and an antiquark. The most common baryons are the proton and the neutron, the building blocks of the atomic nucleus. A great number of hadrons are known, most of them differentiated by their quark content and the properties these constituent quarks confer.
Elementary fermions are grouped into three generations, each comprising two leptons and two quarks. The first generation includes up and down quarks, the second strange and charm quarks, and the third bottom and top quarks. Particles in higher generations generally have greater mass and less stability, causing them to decay into lower-generation particles by means of weak interactions. Only first-generation (up and down) quarks occur commonly in nature. Heavier quarks can only be created in high-energy collisions (such as in those involving cosmic rays), and decay quickly; however, they are thought to have been present during the first fractions of a second after the Big Bang, when the universe was in an extremely hot and dense phase (the quark epoch). Studies of heavier quarks are conducted in artificially created conditions, such as in particle accelerators.
Having electric charge, mass, colour charge, and flavour, quarks are the only known elementary particles that engage in all four fundamental interactions of contemporary physics: electromagnetism, gravitation, strong interaction, and weak interaction. Gravitation is too weak to be relevant to individual particle interactions except at extremes of energy (Planck energy) and distance scales (Planck distance). However, since no successful quantum theory of gravity exists, gravitation is not described by the Standard Model.
As described by quantum chromodynamics, the strong interaction between quarks is mediated by gluons, massless vector gauge bosons. Each gluon carries one colour charge and one anticolour charge. Gluons are constantly exchanged between quarks through a virtual emission and absorption process. When a gluon is transferred between quarks, a colour change occurs in both; for example, if a red quark emits a red-antigreen gluon, it becomes green, and if a green quark absorbs a red-antigreen gluon, it becomes red. Therefore, while each quark’s colour constantly changes, their strong interaction is preserved.
Since gluons carry colour charge, they themselves are able to emit and absorb other gluons. As quarks come closer to each other, the chromodynamic binding force between them weakens; conversely, as the distance between quarks increases, the binding force strengthens. The colour field becomes stressed, much as an elastic band is stressed when stretched, and more gluons of appropriate colour are spontaneously created to strengthen the field. Above a certain energy threshold, pairs of quarks and antiquarks are created. These pairs bind with the quarks being separated, causing new hadrons to form. This phenomenon is known as colour confinement: quarks never appear in isolation. This process of hadronization occurs before quarks, formed in a high energy collision, are able to interact in any other way. The only exception is the top quark, which may decay before it hadronizes.
Hadrons, along with the valence quarks that contribute to their quantum numbers, contain virtual quark-antiquark pairs known as sea quarks. Sea quarks form when a gluon of the hadron’s colour field splits; this process also works in reverse in that the annihilation of two sea quarks produces a gluon. The result is a constant flux of gluon splits and creations colloquially known as “the sea”. Sea quarks are much less stable than their valence counterparts, and they typically annihilate each other within the interior of the hadron. Despite this, sea quarks can hadronize into baryonic or mesonic particles under certain circumstances.
Under sufficiently extreme conditions, quarks may become deconfined and exist as free particles. In the course of asymptotic freedom, the strong interaction becomes weaker at higher temperatures. Eventually, colour confinement would be lost and an extremely hot plasma of freely moving quarks and gluons would be formed. This theoretical phase of matter is called quark-gluon plasma. The exact conditions needed to give rise to this state are unknown and have been the subject of a great deal of speculation and experimentation. A recent estimate puts the needed temperature at 1.90±0.02×1012 kelvin. While a state of entirely free quarks and gluons has never been achieved (despite numerous attempts by CERN in the 1980s and 1990s), recent experiments at the Relativistic Heavy Ion Collider have yielded evidence for liquid-like quark matter exhibiting “nearly perfect” fluid motion.
The quark-gluon plasma would be characterized by a great increase in the number of heavier quark pairs in relation to the number of up and down quark pairs. It is believed that in the period prior to 10−6 seconds after the Big Bang (the ‘quark epoch’), the universe was filled with quark-gluon plasma, as the temperature was too high for hadrons to be stable.
Given sufficiently high baryon densities and relatively low temperatures — possibly comparable to those found in neutron stars — quark matter is expected to degenerate into a ‘Fermi liquid’ of weakly interacting quarks.
So far in this web page we’ve been explaining sub-atomic particles and the way they interact.
And in the article about the Big Bang, we only discovered how hydrogen (and its isotope deuterium) and helium came into existence.
What about all the other elements? Tom Lehrer had a song about them. It doesn’t tell how they came about, but it lists them all with humour. Sky and Telescope magazine from April 2005 has an interesting and authoritative lead [or 82Pb if you prefer‽] into the subject, as does Wikipedia. And here’s a hint of what it’s all about...
In the article about the Big Bang, we discovered how hydrogen (and its isotope deuterium) and helium atoms came into existence some 379,000 years after the Big Bang.
These atoms drifted around, and through gravitational attraction, they formed clumps of atoms; over the years, decades, millennia... these clumps became larger and larger, and, because of the accumulation of irregularities in their motion, they began to rotate. This rotation caused them to form a disk-shaped mass, which gradually shrank because of gravity.
(This account is deliberately abbreviated; whole books have been written on the subject.)
The shrinking disk became denser and hotter at the centre, so hot that eventually nuclear fusion reactions were able to start and a new star was born. The hydrogen and helium were heated and compressed to such an extent that lithium, beryllium, carbon, oxygen, nitrogen, and more of the lighter elements came into existence. (Those protostars with masses less than about 8% of that of the Sun cannot generate the temperatures and pressures necessary for nuclear reactions to occur, and these become brown dwarf stars.)
The fusion reactions were most intense at the centre of the star, where temperatures and pressures became enormous. But all good things must come to an end, and the basic fuel, hydrogen, began to run out. However, the helium continued the process for a time until it was used up.
Two things happened: the centre of the star collapsed forming a white dwarf; and the outer gaseous material exploded outwards to form a nebula. This combined collapse and explosion is called a nova (from the Latin word for ‘new’, because the ancients believed that a new star had been born. Stars with around ten or more times the mass of the Sun can explode in a supernova as their inert iron cores (see below) collapse into an extremely dense neutron star or black hole.
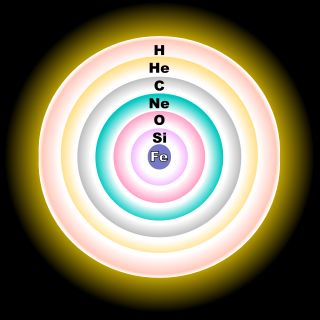
The debris left over from the explosion of the first “generation” of stars, essentially repeats the process just described, with material gradually coalescing into new stars, again with disks. These stars burn hydrogen and helium and some of the newly created elements like carbon, nitrogen and others. This time heavier elements are produced, like iron and lead.
Depending on the mass of the star, different results emerge. Low mass stars may become red dwarfs and survive for thousands of times longer than the Sun (which will exist in its present state for another four billion years). Somewhat larger stars will expand into red giants, while even bigger ones will become supernovae, a truly massive explosion which can completely blow the star apart.
A star with a core mass too great to form a white dwarf but insufficient to achieve sustained conversion of neon to oxygen and magnesium will undergo core collapse before achieving fusion of the heavier elements. Both heating and cooling caused by electron capture onto minor constituent elements (such as aluminium and sodium) prior to collapse may have a significant impact on total energy generation within the star shortly before collapse. This may produce a noticeable effect on the abundance of elements and isotopes ejected in the subsequent supernova.
Once the nucleosynthesis process arrives at iron-56, the continuation of this process consumes energy (the addition of fragments to nuclei releases less energy than required to break them off the parent nuclei). If the mass of the core exceeds the Chandrasekhar limit (about 1½ solar masses), electron degeneracy pressure will be unable to support its weight against the force of gravity, and the core will undergo sudden, catastrophic collapse to form a neutron star or, in the case of cores of neutron stars that exceed the Tolman-Oppenheimer-Volkoff limit (estimated to be between 1½ to 3 solar masses), a black hole.
Each element (except hydrogen-1) contains both protons and neutrons, roughly in equal numbers. The number of protons (and in a neutral atom, the number of electrons in orbit around it) is its atomic number and determines which element it is. So hydrogen always has one proton; if it has one neutron in addition, it is called deuterium, hydrogen-2 or 2H; tritium is hydrogen with two neutrons, 3. Each of these is called an isotope of hydrogen.
14C is a radioactive form of carbon while 12C and 13C are stable isotopes. Radioactivity frequently involves the emission of an alpha-particle, which consists of two protons and two neutrons (a helium-4 nucleus) or a beta-particle (an electron or a positron). In beta decay, a neutron splits into a proton and an electron, and an electron antineutrino is also emitted, while positron emission is accompanied by an electron neutrino. Beta decay is mediated by the weak force. An intermediate W-boson is usually emitted and reabsorbed.
In all cases of such radioactivity, if the proton and neutron are part of an atomic nucleus, these decay processes transmute one chemical element into another. For example,
22Na → 22Ne + e+ + ν+ (beta plus decay; sodium-22 with atomic number 11 becomes neon-22, atomic number 10 and...)
238U → 234Th + 4He (alpha decay; uranium-238, atomic number 92, becomes thorium-234, atomic number 90)
We’ve already met the proton, neutron, electron and their friends; here are some more subatomic particles which either combine to form these or provide the means (forces) by which they are held together. It is important to realise that almost all these particles are extremely short-lived (typically of the order of 10-20 second, and are therefore normally only seen in powerful accelerators (like the Large Hadron Collider) which increase the speed of (typically) protons to something approaching the speed of light and then smash them together. The collisions produce, for a very small amount of time, some of these subatomic particles. It’s this process that is being used to detect the Higgs boson.

